Significance
Point-of-care testing (POCT) is a type of diagnostic testing that is performed outside of the traditional laboratory setting, often at or near the site of patient care. POCT can provide rapid, real-time cost-effective results that can be used to guide clinical decision-making and improve patient outcomes. POCT provides results quickly, often within minutes, allowing for timely intervention and treatment. This is particularly important in emergency or critical care settings, where rapid diagnosis and treatment can be life-saving. Moreover, POCT can help healthcare providers make more informed clinical decisions, leading to improved patient outcomes. For example, rapid diagnosis of a bacterial infection can lead to prompt administration of antibiotics, reducing the risk of complications and improving the patient’s prognosis. Furthermore, POCT allows for testing to be performed at or near the point of care, reducing the need for transportation of specimens to a central laboratory and minimizing the time between specimen collection and test results. It can also be used in settings where traditional laboratory testing may not be available or feasible, such as in remote or resource-limited areas. The technologies used for POCT have seen significant advancements in recent years, with the development of microfluidic devices that use paper or paper-like materials to perform diagnostic tests. These devices are particularly useful in low-resource settings, where access to specialized equipment and trained personnel is limited. However, with the advent of microfluidics in POCT, alternative detection formats are required. This is where fluorescence polarization immunoassay (FPIA) comes into play. FPIA is a widely used technique for measuring the levels of various analytes in a sample. It works by measuring the degree of polarization of a fluorescent signal, which is inversely proportional to the amount of analyte in the sample. The traditional method of performing FPIA is in micro-cuvettes, such as microtiter plates.
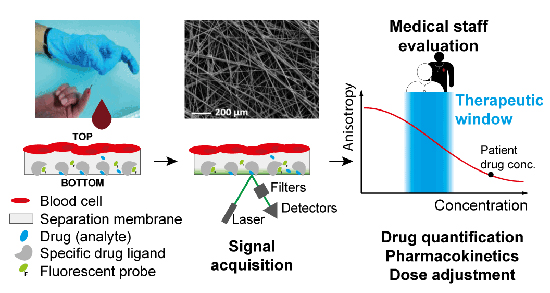
In a new research published in the peer-reviewed journal Analytica Chimica Acta, scientists from the School of Engineering of the University of Applied Sciences Western Switzerland: Dr. E.-Diana Bojescu, Dipl.-Ing. Denis Prim, Dr. Marc E. Pfeifer and Dr. Jean-Manuel Segura, developed a new method for measuring the levels of the antibiotic tobramycin in a patient’s blood using fluorescence polarization immunoassay (FPIA) which was performed within small chambers made of glass fibers.
The authors selected tobramycin to demonstrate the power of their new technology. Tobramycin is an antibiotic medication that is commonly used in medicine to treat a variety of bacterial infections. It belongs to the class of aminoglycoside antibiotics and works by inhibiting bacterial protein synthesis, ultimately leading to bacterial cell death. Monitoring the blood concentration of tobramycin is important because it is a medication that has a narrow therapeutic index, meaning that there is a small difference between the therapeutic dose and the toxic dose. Therefore, maintaining the appropriate blood concentration of tobramycin is crucial to ensure that the drug is effective at treating the bacterial infection while avoiding toxic side effects. When tobramycin is administered, it is distributed throughout the body, including in the kidneys, where it is eliminated from the body. However, in some patients, the drug may not be effectively eliminated, resulting in an accumulation of the drug in the body. This can lead to toxicity, particularly in the kidneys and ears. By monitoring the blood concentration of tobramycin, healthcare professionals can determine whether the drug is being effectively eliminated from the body and adjust the dose accordingly to avoid toxicity. Blood concentration monitoring is typically done by measuring the drug level in the blood at specific intervals after administration, usually every 24 hours.
The research team showed that FPIA can be accurately performed within glass fiber micro-chambers. The use of glass fiber micro-chambers offers several benefits over traditional assay platforms. Firstly, the small size of the chambers increases sensitivity, allowing for the detection of the analyte in minute amounts of blood. Secondly, the micro-chambers can be used to separate plasma from whole blood before quantification, which could prove to be an important asset in the development of solutions for POCT. Lastly, the glass fiber micro-chambers are easy to fabricate and can be incorporated into a small portable device, making it suitable for use in low-resource settings.
The authors demonstrated that this novel combination of glass-fiber chambers with FPIA makes it possible to develop a POCT device for therapeutic drug monitoring using a single drop of blood. The assay could be further simplified by incorporating and drying the reagents within the micro-chambers. Furthermore, the measurement imprecision is very low (CV = 1.2%), which means that the overall analytical performance could be further improved to meet the CLIA criteria and ensure adequate dosage modifications by stabilizing temperature and environmental conditions inside a dedicated FP reader and automating the measurement process. Although POCT devices usually do not reach the precision and accuracy of commercial clinical analyzers, this could be potentially compensated by performing multiple concentration measurements after the drug administration owing to the ease of handling, the rapid turnaround time and the very low blood volume requirements of the FP assay. Thereby more accurate Bayesian estimates of the area under the concentration-time curve (AUC) could be obtained and accurate dose adjustments would be ensured.
In conclusion, this study demonstrates that the use of glass fiber micro-chambers with FPIA is a promising new approach for POCT. The technique is simple, fast, and accurate, and has the potential to be used in therapeutic drug monitoring at the point of care, particularly in low-resource settings. This technique will help healthcare professionals to monitor blood concentration of tobramycin for example and detect early signs of toxicity and intervene as necessary.
Reference
Bojescu ED, Prim D, Pfeifer ME, Segura JM. Fluorescence-polarization immunoassays within glass fiber micro-chambers enable tobramycin quantification in whole blood for therapeutic drug monitoring at the point of care. Analytica Chimica Acta. 2022;1225:340240.
 Medicine Innovates Medicine Innovates: Delivering innovations in medicine to the world for better health and prosperity
Medicine Innovates Medicine Innovates: Delivering innovations in medicine to the world for better health and prosperity
