Significance
Scientists from the University of Cambridge have developed a highly porous type of nanoparticle known as a metal-organic framework (MOF) that can deliver small interfering RNAs (siRNAs) directly to cells. There is considerable interest in using siRNAs to knock down disease-related genes as an approach to cancer therapy, but delivering the molecules to cells intact is challenging. The Cambridge University-led researchers, headed by David Fairen-Jimenez, PhD, at the department of chemical engineering and biotechnology, used computational simulations to find a MOF with the ideal pore size, which could encapsulate and carry siRNA molecules to target cells, and then break down and release its siRNA cargo once inside the cells. Their research work is now published in Chem.
siRNAs are small, double-stranded RNA fragments, typically 21 to 23 nucleotides in length, which can be designed to block the expression of specific genes and so prevent their proteins from being produced. The technology holds great potential as a therapeutic approach against hard-to-treat cancers, as well as other neurological disorders and viral infections, the authors noted. siRNAs are particularly attractive as a strategy for blocking over-expressed cancer-driving genes because the molecules are highly efficient at gene knockdown, but they are also nontoxic, highly specific—which minimizes the risk of off-target effects—and don’t trigger immune responses. All that is needed to design a siRNA is the sequence of the gene that is targeted for knockdown. And, unlike traditional drug design strategies that are based on expensive and time-consuming rounds of synthesizing and testing new drugs for each condition, redesigning an siRNA to target a different gene is relatively simple.
One of the main limiting factors in developing siRNAs as viable drug to treat disease is that the molecules are very unstable and are often broken down before they can reach their targets. “Although siRNA therapy has the potential to benefit patients with cancer, the main limitation is its lack of stability and ease of degradation by native biological enzymes,” the investigators wrote. Chemically modifying the siRNA molecules can make them more stable, but this can compromise their ability to knock down the target genes. It’s also difficult to get the siRNA molecules into cells. They need to be transported by another vehicle acting as a delivery agent.
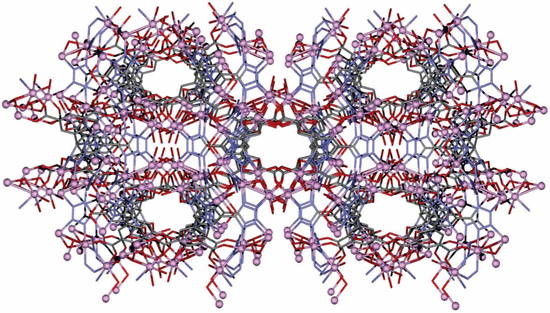
The research team who are experienced in MOFs, (self-assembling 3D compounds made of metallic and organic building blocks connected together). There are thousands of different types of MOFs that researchers can synthesize, including more than 84,000 MOF structures currently in the Cambridge Structural Database—to which 1,000 new structures are published each month. The properties of MOFs can also be fine-tuned for specific purposes. By changing different components of the MOF structure, researchers can create molecules with different pore sizes, stabilities, and toxicities, including structures that can carry molecules such as siRNAs into cells without harmful side effects.
For siRNA delivery, the MOF carrier needs to be biodegradable so that it doesn’t then build up in cells. The MOF that authors selected (zirconium-based metal node ) breaks down into harmless components that are easily recycled by the cell without toxic side effects. The large pore size of the MOF designed by the team also means that a significant amount of siRNA can be loaded into each MOF carrier, so the dosage needed can be kept very low. To evade endosomal entrapment, the authors engineered MOF with various cofactors (proton sponge, KALA peptide, and NH4Cl to ensure optimal gene knockdown.
The versatility of the system designed will enable the use the same adapted MOF to deliver different siRNA sequences and target different genes. Because of its large pore size, the MOF also has the potential to deliver multiple drugs at once, which could lead to the development of combination therapies.
Reference
Michelle H.Teplensky et al. A Highly Porous Metal-Organic Framework System to Deliver Payloads for Gene Knockdown. Chem. Available online 16 September 2019.
 Medicine Innovates Medicine Innovates: Delivering innovations in medicine to the world for better health and prosperity
Medicine Innovates Medicine Innovates: Delivering innovations in medicine to the world for better health and prosperity

